LMT Communications, Inc. recently posted an article featuring 5 dental lab professionals and their takeaways from operating during the global pandemic. Hearing what other dental labs have experienced through the coronavirus threat, and how they’ve learned to overcome the unprecedented challenges that came with it, is very helpful in figuring out how to you can safeguard your lab from unforeseen circumstances in the future.
Atlanta Based Systems has lead the industry with our state-of-the-art dental lab management software for over 40 years. With Evolution, your lab can streamline your workloads and stay lean. The features available within the applications provided by ABS in our Evolution dental lab management software can give you tools to manage cases currently in production, as well as insight on what’s to come for your lab.
One of the dental lab professionals in LMT’s article mentions having to lay off his assistant and consolidate their department managers to one operations manager responsible for all of their departments. With EvoMGR from ABS, lab or department managers can keep track of the cases being processed through their departments and have the ability to change schedules, reassign techs, add notes/images and much more.

Another dental lab professional featured in the article talks about “cross training” employees so that “each department could operate independently.” In Evolution, User profiles can be created or edited easily giving access to different parts of the system allowing for certain employees to multitask their case management and customer service.
At the end of the article, Russellville Dental Lab Managing Partner, Lee Coursey, discusses watching the right metrics:
“We realized it didn’t make sense to base it on sales because—since that work is already done and out the door—we’d be making decisions five to 10 days behind. Instead, we focused on incoming workloads: how much there was and how long would it be in-house. Shifting our focus to the throughput—the number of units we would be moving through each department every day—helped us gain clarity.”
Mr. Coursey has been using Evolution and working with Atlanta Based Systems for many years. The tools and features that Evolution offers were able to help Lee extrapolate relevant metrics needed to help them develop a plan for layoffs based on historical workloads.
If you’re interested in ABS and getting a personal demonstration of Evolution, please give us a call or drop us a line.



 In March, The New York Times listed Dentistry as one of the professions at highest-risk of the novel coronavirus infection. In light of the study, the vice president of the ADA Health Policy Institute, Marko Vujicic, Ph.D., said, “The fact that dentistry was named one of the most at-risk professions for infection, but has a far lower prevalence of infection compared to other health professions, is not a coincidence. The profession has taken this issue extremely seriously, and it shows.”
In March, The New York Times listed Dentistry as one of the professions at highest-risk of the novel coronavirus infection. In light of the study, the vice president of the ADA Health Policy Institute, Marko Vujicic, Ph.D., said, “The fact that dentistry was named one of the most at-risk professions for infection, but has a far lower prevalence of infection compared to other health professions, is not a coincidence. The profession has taken this issue extremely seriously, and it shows.”
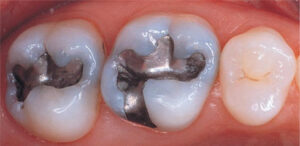 Although the FDA defines these groups as ‘maybe’ having a higher-risk of negative health effects from dental amalgam, they don’t appear to provide any new evidence of this being the case. In fact, after the FDA released their statement, the ADA (American Dental Association) released their own press release reaffirming its position on dental amalgam.
Although the FDA defines these groups as ‘maybe’ having a higher-risk of negative health effects from dental amalgam, they don’t appear to provide any new evidence of this being the case. In fact, after the FDA released their statement, the ADA (American Dental Association) released their own press release reaffirming its position on dental amalgam.